
Feldspar Powder Manufacturer, Supplier & Exporter in Rajasthan, India
WHAT IS IT?
Feldspar comprises of group of aluminum silicate that contains sodium (NaAlSi3O8), potassium (KAlSi3O8)or calcium(CaAl2Si2O8).Feldspars are the most abundant mineral in Earth’s crust, making up more than 60% of the crust.

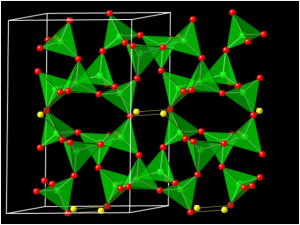
MOLECULAR STRUCTURE
The basic unit of Feldspar Powder structure is a tetrahedron of silicon and oxygen (SiO4) and aluminium and oxygen (AlO4–). These tetrahedra are covalently bonded to each other through oxygen atoms to form a three dimensional framework. Low-charge cations like Na+, K+ or Ca2+ occupy the larger voids in the tetrahedral framework to provide charge balance against the aluminium tetrahedra.
PROPERTIES
1.Tactile properties: It is rough and abrasive.
2.Visual properties:
- Color: It is white in color but is also found in various shades like pink, brown etc, depending upon the impurities present. Color is white or none, though shades of gray, red, orange, yellow, green, blue, violet, brown, or even black can occur when the mineral is charged with impurities.
- Luster: It’s luster is vitreous to pearly on cleavage surfaces in crystallized varieties and vitreous to resinous to dull in massive forms.
- Streak: It’s streak is white.
- Diaphaneity: It is transparent to opaque.
- Hardness: 6-6.5 mohs.
- Specific gravity:2.6
4.Affinity to water: It dissolved slightly in water, releasing the alkali metal ions.
5.Affinity to oil: It has low oil absorption capacity.
6.Heat properties:
- Thermal conductivity: 2.3-2.5 W/m/K.
- Specific heat capacity: 222 J/M/K.
- Coefficient of expansion: 13.1 to 30.6.
- Melting point: 1200 to 1400 °C.
- Decomposition temperature: 1473K
8.Combustion properties: It is non explosive and non inflammable.
9.Chemical properties: It dissolves in acid, releasing the alkali metal ion and aluminium ion.
