
Dolomite Powder Manufacturer In India
WHAT IS IT?
It is a naturally occurring mineral CaMg(CO3)2.

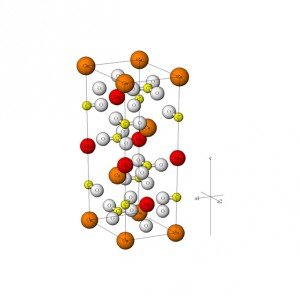
MOLECULAR STRUCTURE
Dolomite structure is formed by an ionic interaction of Mg2+ (magnesium cation), Ca2+ (calcium cation), and CO32- (carbonate anion). For every two carbonate anions there is one magnesium cation and one calcium cation, thereby making the structure electrically neutral. A common pattern of arrangement of ions is shown:
PROPERTIES
1.Tactile properties: It is slightly rough and abrasive.
2.Visual properties:
- Color: It is often in pink or pinkish and can be colorless, white, yellow, gray or even brown or black when iron is present in the crystal white, grey, pink color.
- Lustre: It is vitreous to pearly to dull.
- Streak: It is white.
- Diaphaneity: It is opaque.
- Hardness: 3.5-4 mohs.
- Specific gravity: 2.84-2.86.
5.Affinity to oil: It has low oil absorption capacity.
6.Heat properties:
- Thermal conductivity: 5.1 W m-1 K-1 at 20 °C, It exhibits a two stage decomposition (750 ° Dolomite and MgO; 900 ° CaO and MgO).
- Specific heat capacity: 900 J kg-1 K-1 at 20 °C.
- Mean thermal expansion coefficient: 4.1×10-6/°C.
- Melting Point: 2570 °C.
8.Combustion properties: It is non explosive and non-inflammable.
9.Chemical properties: It’s effervesces is weak with warm acid or when first powdered, with cold HCl.
APPLICATIONS
Theoretically, Dolomite contains 54.35% CaCO3 and 45.65% MgCO3. Its composition can also be expressed in terms of the oxides. Thus, in other words, it contains 30.4% CaO, 21.7% MgO and 47.9% CO2.
However, in nature considerable variations arise in dolomite due to the following factors:
- Varying proportions of lime and magnesia.
- Presence of impurities, chiefly silica, alumina and iron oxide.
- Differences in geological formations.
