
Pyrophyllite Powder Manufacturer, Supplier & Exporter in Rajasthan, India
WHAT IS IT?
Pyrophyllites a naturally occurring mineral. Its chemical name is hydrated aluminium silicate AlSi2O5OH or Al2O3.4SiO2.H2O. Thus, it contains 28.3% Al2O3, 66.7% SiO2 and 5% H2O.

MOLECULAR STRUCTURE
Pyrophyllite Powder structure consists of two Phyllosilicate sheets. These are silica tetrahedra (SiO4) covalently bonded by sharing oxygen atoms as shown below to form a sheet. The sheet thus formed has a ring like arrangement of tetrahedral, each ring containing 6 tetrahedra.

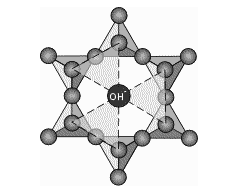
Three oxygen per tetrahedron satisfy their valencies by covalently bonding to other tetrahedra in forming the sheet. The oxygen atoms at the tips of the sheet are significant as they do not interact with other oxygen atoms. It therefore has a gained electron to complete its shell and one negative charge. In the center of each silicate ring is an OH- ion as shown below.
The OH- ion is on the same plane as the tip oxygen. Now imagine such sheets one on top of the other and consider the circled portion.
- 2 tip oxygen of the sheet above (O-).
- 2 tips oxygen of the sheet below (O-).
- The OH- of the sheet above.
- The OH- of the sheet below.

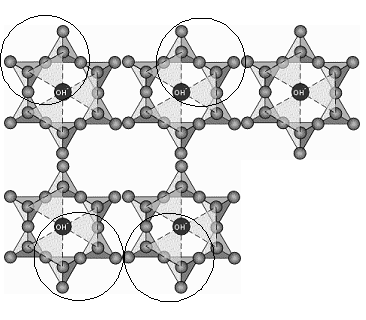
All connect with each other to form an octahedron. In the center of this octahedron aluminium cation is placed.Similar octahedral are formed in every unit.
Thus effectively, every unit (the circled portion + the corresponding portion in the sheet below), ends up forming 2 octahedra. Thus, for every:- 2 tip oxygens of the sheet above (O-)
- 2 tips oxygens of the sheet below (O-)
- The OH- of the sheet above
- The OH- of the sheet below
PROPERTIES
1.Tactile properties: It is smooth, slippery, soapy and greasy.
2.Visual properties:
- Color: It is in green, yellow or white often with a brown or grey hue color.
- Lustre: It is dull and pearly.
- Diaphaneity: It is translucent to opaque.
- Hardness: 1.5-2 mohs
- Specific gravity: 2.65 to 2.9
5.Affinity to oil: Pyrophyllite can absorb oil between its molecular layers. However, the oil absorption capacity can vary from grade to grade.
6.Heat properties:
- Thermal conductivity: 0. 04 j/cm/s/K along the direction of the layers and half this value perpendicular to the layers.
- Cp: 320.5 J/M/K.
- Expansion coefficient: 14.9/K in one direction and 12.2/K in the perpendicular direction.
- Starts decomposing at 650 to 850 °C.
- Melting Point : It melts slowly over wide temperature range (1200 to 1630 °C).
8.Combustion properties: It is non explosive and non inflammable.
9.Chemical properties:
- Neutral
